 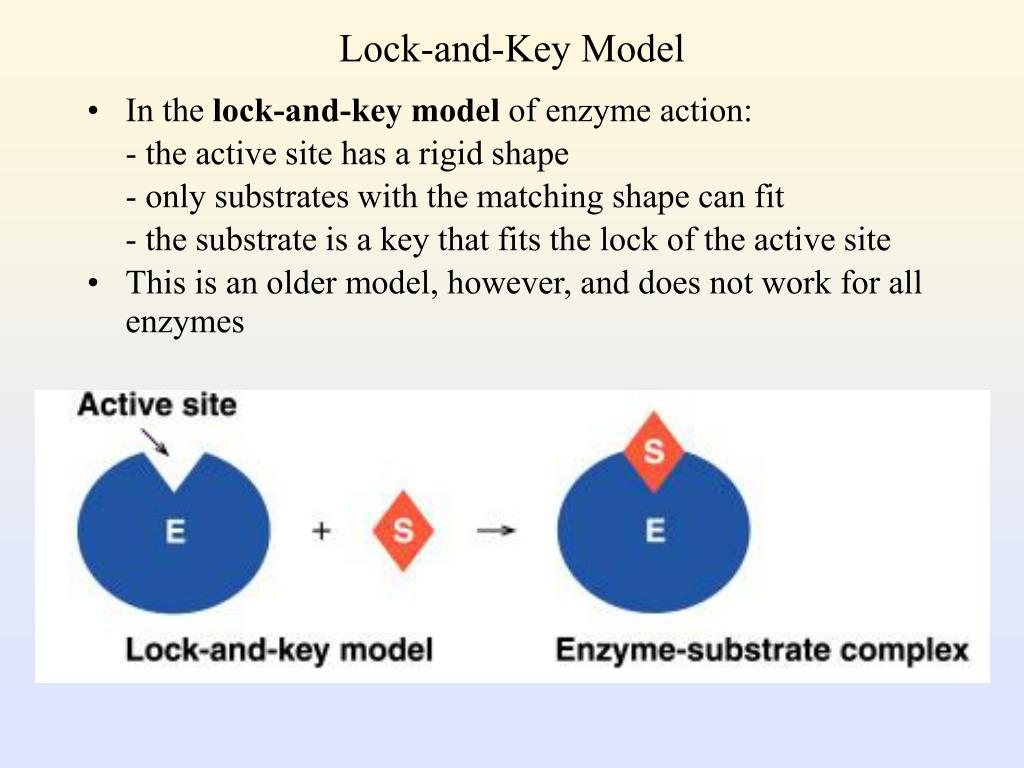
This explains enzyme specificity This explains the loss of activity when enzymes denature Simple account of how enzymes work: © 2007 Paul Billiet ODWSĦ Denaturation Heat proteins above 40oC (or treat with certain chemicals or radiation) they will gradually loose their 3D shape Ability to react with substrate is lost This change in shape and loss of biological activity is called DENATURATION Normally permanent process The substrate no longer fits the active site, so that is not converted into productħ Task-2 Why is enzyme specificity is a key feature in enzyme action?Įxplain difference between extra cellular and intra cellular enzymes with 4 examples each. Temporary structure called the enzyme-substrate complex formed Products have a different shape from the substrate Once formed, they are released from the active site Leaving it free to become attached to another substrate © 2007 Paul Billiet ODWSĪn enzyme binds a substrate in a region called the active site Only certain substrates can fit the active site Amino acid R groups in the active site help substrate bind Enzyme-substrate complex forms Substrate reacts to form product Product is releasedĮnzyme may be used again Enzyme-substrate complex E S P Reaction coordinate © 2007 Paul Billiet ODWSĤ Lock and Key Model E S ES complex E P P S S P For example, intestinal enzymes have an optimum pH of about 7.5, but stomach enzymes have an optimum pH of about 2.Fit between the substrate and the active site of the enzyme is exact Like a key fits into a lock very precisely The key is analogous to the enzyme and the substrate analogous to the lock. The optimum pH for an enzyme depends on where it normally works. Different enzymes work best at different pH values. 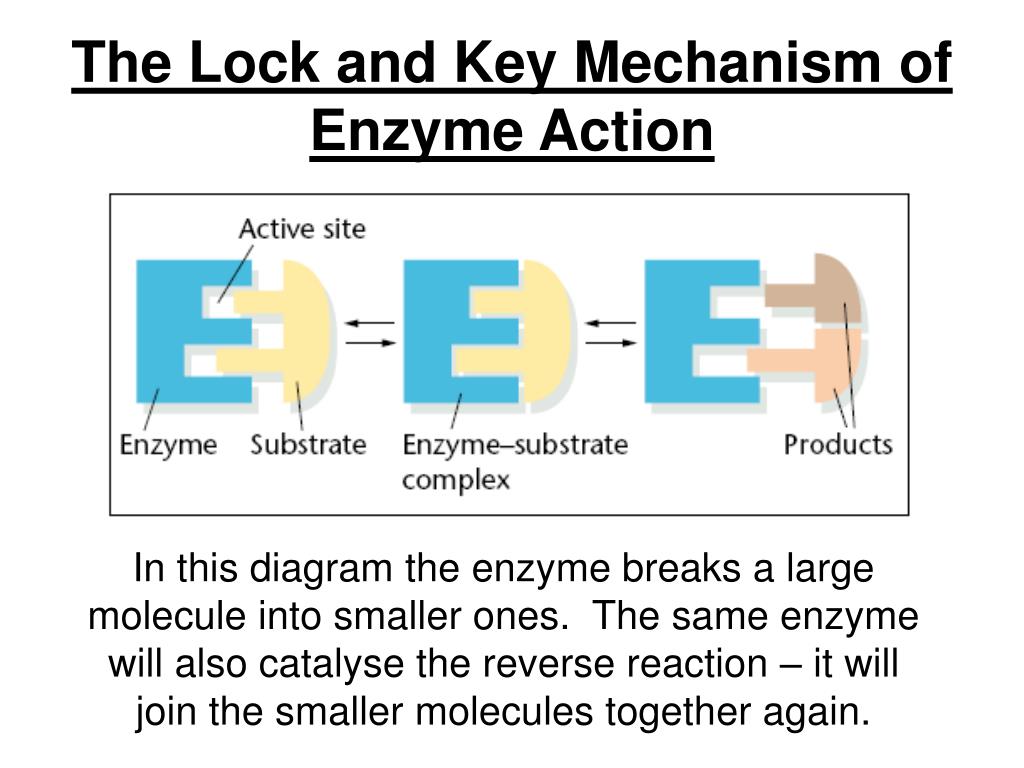
The enzyme is denatured.Ĭhanges in pH alter the shape of an enzyme’s active site. Enzyme–substrate complexes can no longer form as the substrates no longer fit into the active site. If the temperature continues to increase past the optimum, the increased kinetic energy breaks the weak hydrogen bonds holding the enzyme’s unique active site shape.At the optimum temperature the maximum number of enzyme-substrate complexes form per unit time.As the temperature increases, the kinetic energy increases, leading to more collisions and enzyme substrate complexes formed per unit time.This results in the particles colliding less often, which means there will be fewer successful collisions between the substrate and the enzyme’s active site. At low temperatures the enzymes and substrates have low kinetic energy.However, at high temperatures the rate decreases again because the enzyme becomes denatured and can no longer function as a biological catalyst. Effect of temperatureĪs with ordinary chemical reactions, the rate of an enzyme-catalysed reaction increases as the temperature increases. Enzymes can be denatured by high temperatures or extremes of pH. We say that the enzyme has been denatured. 
If the shape of the enzyme changes, its active site may no longer work. In this example, the enzyme splits one molecule into two smaller ones, but other enzymes join small molecules together to make a larger one. This makes enzymes highly specific – each type of enzyme can catalyse only one type of reaction (or just a few types of reactions). In the lock and key model, the shape of the active site matches the shape of its substrate molecules. The place where these molecules fit is called the active site. Lock and key modelĮnzymes are folded into complex shapes that allow smaller molecules to fit into them. Enzymes have active sites which only match specific substrates. Structure of enzymes – Higher tier onlyĭifferent enzymes contain up to 20 different amino acids linked together to form a chain which then folds into the globular enzyme shape. An enzyme is a protein that functions as a biological catalyst – a substance that speeds up a chemical reaction without being changed by the reaction. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
