 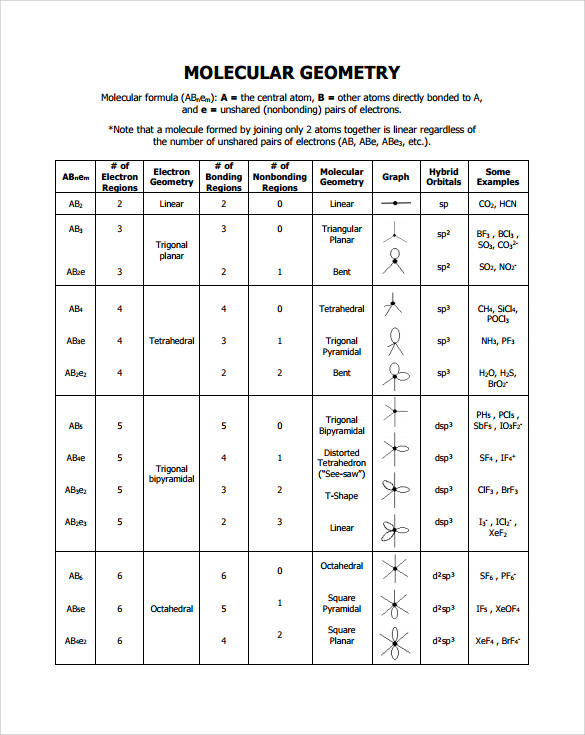
It is trigonal pyramidal because it is attached to four "things": the three hydrogens and a non-bonding pair of electrons (to fulfill nitrogen's octet).Ĭonsider a simple covalent molecule, methane (CH 4). Ammonia (NH 3) is not trigonal planar, however. For example, methane (CH 4) is tetrahedral-shaped because the carbon is attached to four hydrogens. The number of groups is how many atoms or electron pairs are bonded to the central atom. Those "things" can be other atoms or non-bonding pairs of electrons. The hybridization is determined by how many "things" are attached to the central atom. Table of Geometries Orbital Hybridization Modern methods of quantitatively calculating the most stable (lowest energy) shapes of molecules can take several hours of supercomputer time, and is the domain of computational chemistry. This can allow us to predict the geometry of similar molecules, making it a fairly useful model. The VSEPR model is by no means a perfect model of molecular shape! It is simply a system which explains the known shapes of molecular geometry as discovered by experiment. Wikipedia has related information at VSEPR Within this model, the AXE method is used in determining molecular geometry by counting the numbers of electrons and bonds related to the center atom(s) of the molecule. A scientific model, called the VSEPR (valence shell electron pair repulsion) model can be used to qualitatively predict the shapes of molecules. There are several different methods of determining molecular geometry. This repulsion causes covalent molecules to have distinctive shapes, known as the molecule's molecular geometry. 
Being mutually negatively charged, the electron pairs repel the other electron pairs and attempt to move as far apart as possible in order to stabilize the molecule. Covalent molecules are bonded to other atoms by electron pairs. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
